
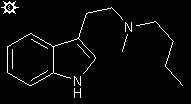
#27. MBT
|
| [3D .mol Image] |
A stirred suspension of 6.3 g of N-butyl-N-methyl-indoleglyoxylamide in 150 mL dry toluene, in a three-neck round bottomed flask and under a N2 atmosphere, was cooled with an external ice bath. A total of 30 mL of a 65% RED-AL solution in toluene was added slowly by syringe, and there was immediate gas evolution. After the addition was complete, the stirring was continued for an hour, then the flask was gradually warmed to 40 °C, and the stirring continued for an additional 2 h. After cooling again to ice temperature, the excess RED-AL was decomposed by the drop-wise addition of first IPA followed by (after conspicuous gas evolution had ceased) H2O. The inorganic aluminum salts were removed by filtration of the resulting suspension, and the filter cake was washed with isopropyl acetate. The filtrate and washings were combined and washed thoroughly with H2O. The product was then extracted into 1 N hydrochloric acid, the pooled extracts washed twice with CH2Cl2, made basic with 20% aqueous KOH and extracted with CH2Cl2. After washing with H2O and drying with anhydrous Na2SO4, the solvent was removed under vacuum to yield a light yellow oil with a bluish florescence. The free amine did not crystallize but was dissolved in MeOH and titration to a slightly basic end-point with a methanolic solution of fumaric acid. The solution was heated to the boiling point, and slowly diluted with two volumes of hot isopropyl acetate. Slow cooling yielded beautiful light yellow crystals of N-butyl-N-methyl-tryptamine fumarate (MBT). A recrystallization from MeOH/isopropyl acetate gave 5.83 g (69%) of product with a mp 148-150 °C.
DOSAGE : 250 - 400 mg
DURATION : 4 - 6 hrs
QUALITATIVE COMMENTS : (with 130 mg, orally) "Perhaps a subtle intoxication at two hours, and certainly nothing at five hours."
(with 175 mg, orally) "Some mild incoordination and concentration difficulties, all trivial, and a good sleep and a good day the next day."
(with 250 mg, orally) "At 75 minutes there was the prompt development of an intoxicated state primarily characterized by fine motor impairment. Nothing remotely resembling any type of hallucination. Appetite was normal and food and water were consumed without difficulty. Most activities were uninteresting, even dull. The effects lasted about five hours."
(with 400 mg, orally) "It hit an just over an hour, and it quickly became difficult to keep both eyes focused on the point of gaze. There was no actual double vision, but things were not quite right. In a few more minutes an apparent motion became apparent with fixed objects, and shortly thereafter there was a faint 'retinal circus' that was reminiscent of DMT but less compelling. Subject matter could not be chose, but rather came on its own. At this point, walking required great concentration, and lying on a bed was a much better choice. Music seemed to encourage the drifting of thoughts, but all the eyes-closed effects faded quite quickly. I felt overheated, sweat a lot, was intensely dehydrated, and drank quantities of water all night, and still felt dehydrated. Urine output was low. Not my choice of drug; the intoxication is too much for the visual stuff."
EXTENSIONS AND COMMENTARY : This is a pretty heavy body trip for a modest mental return. As with any tertiary amine, one cannot help but speculate what role the deamination enzyme systems of the liver are compromising the results that are being experienced. Here is a compound with five aliphatic carbon atoms hanging out there on the basic nitrogen. No branched chain; everything straight chain. How does this be compared with other tryptamines with straight-chain on that nitrogen atom? DMT has two such carbon atoms. DET has four, DPT has six and DBT has eight. With a tally of five, MBT should lie in-between DET and DPT. Both of these show oral activity in the 300 milligram range (as does MBT) but at least DET has some five-fold increased potency if given parenterally. I really would like to see this particular compound explored by smoking, or injection, or even orally but with some effective monoamine oxidase inhibitor on board (a dose of P. harmala maybe) and see if there can be more mental effects and fewer toxic effects at a lower dose exposure. There is certainly good precedent for it, amongst the other dialkyl tryptamines.
A structural isomer has been made, with the butyl group branched at the nitrogen atom. This is N-s-butyl-N-methyltryptamine, or MSBT. It came from a two pass alkylation of N-methyltryptamine (NMT) with s-butyl bromide in isopropylalcohol in the presence of solid potassium iodide. It remained an oil, but was over 90% pure by GCMS, with unreacted NMT being the major impurity. MS (in m/z): C6H14N+ 100 (100%); indolemethylene+ 130 (8%); parent ion 230 (1%). It has been assayed in man, but it remains an unknown.