
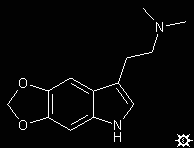
#31. 5,6-MDO-DMT
TRYPTAMINE, N,N-DIMETHYL-5,6-METHYLENEDIOXY; INDOLE, 3-[2-(DIMETHYLAMINO)ETHYL]-5,6-METHYLENEDIOXY; N,N-DIMETHYL-5,6-METHYLENEDIOXYTRYPTAMINE; 3-[2-(DIMETHYLAMINO)ETHYL]-5,6-METHYLENEDIOXYINDOLE; 5H-1,3-DIOXOLO-[4,5-F]INDOLE-7-ETHANEAMINE, N,N-DIMETHYL
|
| [3D .mol Image] |
To a well-stirred suspension of 0.77 g of LAH in 40 mL dry THF, there was added dropwise a solution of 0.87 g 5,6-methylenedioxy-N,N-dimethylglyoxylamide in approximately 100 mL of anhydrous THF. The mixture was brought to reflux temperature, held there for 2 h, and allowed to return to room temperature. It was hydrolyzed by the cautious addition of 0.8 mL H2O, followed with 2.4 mL 10% aqueous NaOH, and finally an additional 0.8 mL of H2O. The inorganics were removed by filtration through Celite, and the filtercake was washed with additional THF. After removal of the solvent of the combined filtrate and washings under vacuum, the residue was distilled by KugelRohr and the colorless distillate recrystallized from a mixture of EtOAc / hexane. There was thus obtained 0.30 g 5,6-methylenedioxy-N,N-dimethyltryptamine (5,6-MDO-DMT), mp 115-117 °C (yield 38%). Anal: C,H,N.
DOSAGE : Greater than 5 mg
DURATION : unknown
QUALITATIVE COMMENTS : (with 5 mg, smoked) "Nothing."
EXTENSIONS AND COMMENTARY : Up until 15 years ago, there had been no research published describing any simple N,N-disubstituted tryptamines carrying the methylenedioxy substitution pattern on the indole ring. This is interesting in that the activity of the methylenedioxy substituted phenethylamine MDA had been well documented almost 40 years ago, and its N-methyl homologue MDMA has been of known human activity for about 25 years. Then, within a year, two papers appeared in the literature describing both this compound (5,6-MDO-DMT) and the corresponding N,N-diisopropyl homologue, 5,6-MDO-DIPT. As to the position of this five-membered ring, there are two appealing locations. The 5,6-pattern has an appealing symmetry to it, being closely parallel to MDA with a sort-of long axis extending through the tryptamine molecule from the 3-position (where the side chain is attached) across the indole ring coming out between the 5- and 6- positions. This certainly feels like the most natural analogue to MDA or MDMA. It has the plus of having the important 5-position occupied, but there might be a bit of a negative effect due to its having something at the 6-position. A more exciting possibility would be the 4,5-disubstitution, which would involve the favorite 5-position along with the site of the oxygen atom of psilocin and psilocybin. This compound has been made and is the preceding recipe, 4,5-MDO-DMT.
As to the nature of the nitrogen substituents, this N,N-dimethyl compound is directly analogous to its 5-methoxy or its 5-hydrogen counterparts. In behavioral disruption studies, it is less potent than either of the simpler compounds, 5-MeO-DMT or DMT. In human studies these latter two chemicals are both active at levels of a few milligrams, and a trial with 5,6-MDO-DMT showed no activity at all at a five milligram trial. More studies are needed, and I am sure that, in time, they will be carried out.